Methods for Acoustic Detection of Insect Pests in Soil
Richard W. Mankin1, Clayton W. McCoy2, Kathy L. Flanders3, Jamee Brandhorst3, Robert L. Crocker4, and Jeffrey P. Shapiro1
1United States Department of Agriculture, Agricultural Research Service, Center for Medical, Agricultural, and Veterinary Entomology, Gainesville, FL 32604, 2University of Florida Citrus Research and Education Center, Lake Alfred, FL 33850, 3Department of Entomology, Auburn University, Auburn, AL 36849, 4Texas Agricultural and Mechanical University Research and Extension Center, Dallas, TX 75252
ABSTRACT
There is considerable practical need for userfriendly, inexpensive devices that detect and quantify underground insect populations. One approach used with varying success is to detect the insects through the sounds or vibrations they generate for communication or through noises that are produced incidentally during feeding and general movement. We have conducted field tests with portable accelerometer and microphone systems that can detect Phyllophaga white grubs and Diaprepes citrus root weevils at distances of 20 50 cm. The ability to precisely monitor soil insect populations and identify specific pests is limited by the high rate of attenuation of sound in soil and a lack of speciesspecific features in the typically broadband spectrum of movementgenerated signals. However, we have had success at generating spectral templates that can distinguish many insect sounds from those produced by vehicles, wind, and other lowfrequency noise generators.
INTRODUCTION
Soildwelling insect pests cause billions of dollars of damage yearly to turf, agricultural crops, trees, and golf courses (1,2), but their infestations are difficult to control because the immature grubs that cause most of the damage are hidden from view. Growers and managers need reliable, inexpensive tools to target infested sites, eliminate costly treatment of uninfested areas, and reduce management costs. Researchers need to develop more knowledge about life cycles, behavior, and population distributions, and determine the efficacy of new management strategies. The use of acoustic sensors and digital signal processing techniques provides a means of identifying and targeting populations that are monitored now only by laborious, destructive techniques.
The usefulness of acoustic techniques for detection of soil insects depends on several biophysical factors, including the signaltonoise ratio, the amount of distortion and attenuation as the sounds travel through the soil, the distinguishability of sounds made by other organisms, and the fraction of the measurement period during which signals are generated. Sound attenuation is greater in soil than in air (~600 dB m-1, compared with ~0.008 dB m-1, 3, 4). However, there is evidence that low frequency sounds (<5 kHz) traveling through sandy soil can be detected over distances of 550 cm, and detectable sound transmission has been reported through plants and leaf mats for distances up to 8 m (5).
We have conducted studies at several sites in Florida and Alabama with portable accelerometer and soil microphone systems to determine the detection range of acoustic sensors and identify particular signal features that can be used to distinguish pest from nonpest organisms in the field. This report describes some initial results of analyses to develop spectral and temporal templates to distinguish between sounds made by Phyllophaga (white grubs), Diaprepes (citrus root weevils), Scapteriscus (mole cricket), and other pest insects, and extraneous background noises (airplanes or automobiles) or sounds made by nonpest insect organisms (e.g., millipedes, earthworms, and earwigs).
METHODS
Accelerometer. Insect sounds were recorded from soil under orange trees in an experimental grove at the University of Florida Citrus Research and Education Center, Lake Alfred, FL. Groups of trees were seeded over an 8week period with Diaprepes abbreviatus eggs or neonates reared at the USDAARS Horticultural Research Laboratory, Orlando, FL, and other trees were left uninfested. Sounds from infested and uninfested trees were recorded 6 times over a 3month period during and after seeding. The presence or absence of live D. abbreviatus larvae was confirmed for 10 trees at the end of the final recording session by pulling the trees, shaking the soil from the roots, and sieving the soil.
To record, 30cmlong steel spikes or 3 × 31.5 × 0.5cm steel stakes were driven into the soil under the drip line of the tree canopy. An accelerometer (Brüel and Kjær [B&K] model 4370, Nærum, Denmark) was attached magnetically ~ 5 cm above the soil surface. Signals collected for 3min periods were passed to a B&K model 2635 charge amplifier and stored on a portable digital audio tape recorder (TEAC model DAP1, Montebello, CA). Afterwards, the recorded signals were amplified 1020 dB with a B&K Model 2610 amplifier, bandpassed filtered between 23000 Hz (KrohnHite model 3100, Avon, MA), digitized at 25 kHz, and analyzed using customwritten software (6).
Spectral templates were constructed by listening to recordings from trees where D. abbreviatus had been sieved and averaging spectrum levels in 164ms intervals (4096 digitized points) where detectable sounds were produced. Typical sounds were 2200 ms in duration. 100-200 sounds were averaged to produce each template. Accelerometer vibration levels were measured in dB re 10-6 ms-2.
Soil Microphone. Turf pest insect sounds were recorded in hay fields at Clarke County, AL, and mole cricket sounds were recorded by Jamee Brandhorst at the Red Eagle Golf Course at Eufaula, AL using a portable soil microphone system developed by Robert Hickling at the National Center for Physical Acoustics. The soil microphone had a broader range of sensitivity than the accelerometer and was encased in a rugged housing. After recordings at the Clarke county hay field, the soil within a 20cm radius of the microphone was dug up with a shovel examined for visible organisms by hand. Recording, digitization, and analysis procedures were the same as for the accelerometer except that the pass band of the filtering was 10010,000 Hz. Templates were constructed using the procedures described above from recordings collected at sites where soil cores had revealed only white grubs, earthworms, earwigs, millipedes, carabid beetles, or ants. Incidental background sounds including wind and motor vehicle noise were identified by listeners, and templates were constructed for these sounds also. At the Red Eagle golf course, mole cricket templates were constructed from recordings at sites where adult mole crickets were observed. 30200 sounds were used in the construction of each of the soil microphone templates.
RESULTS AND DISCUSSION
Accelerometer. The portable accelerometer system detected D. abbreviatus feeding and movement sounds at several recording sites under orange trees in the Lake Alfred grove (Fig. 1).

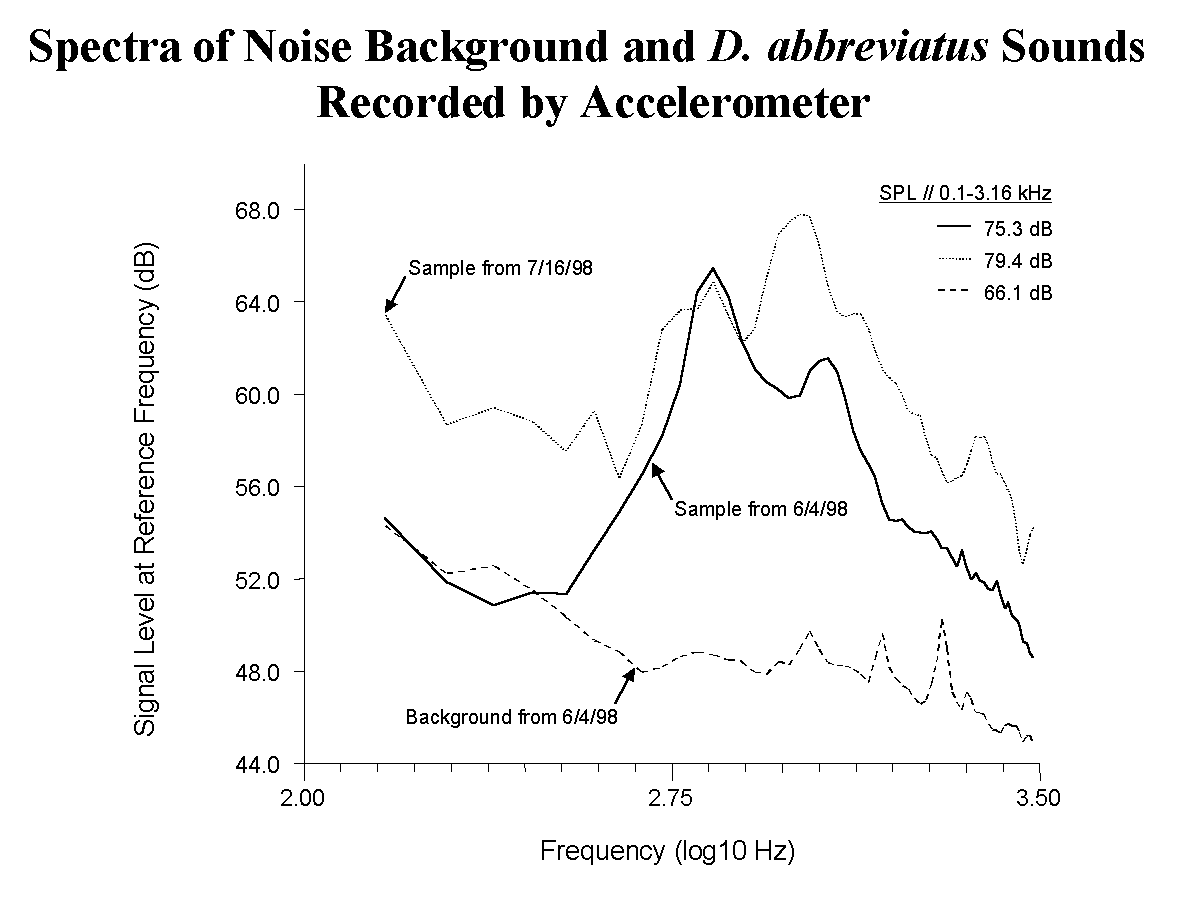
Figure 1. Spectra of background sounds and D. abbreviatus sounds averaged from two different recording sessions with the accelerometer system in an orange grove.
Typically, 50250 sounds were recorded over a 3min period when the insects were present. Samples of recordings averaged at two sites on two different days had sound pressure levels of 7579 dB between 0.1 and 3.16 kHz, with two prominent peaks near 0.7 and 1.1 kHz. Either or both peaks can be used to distinguish the D. abbreviatus signals from extraneous background noises with peak spectrum levels below 300 Hz. Background levels were about 65 dB. Soil samples were relatively free of other large, soildwelling organisms except for ants.
Soil Microphone. The sounds produced by the turf insect pests (Phyllophaga) in the Clarke County hay field could easily be distinguished from a quiet background because there were significant, broadband contributions to their signals above 400 Hz that were lacking in the background noise (Fig. 2).
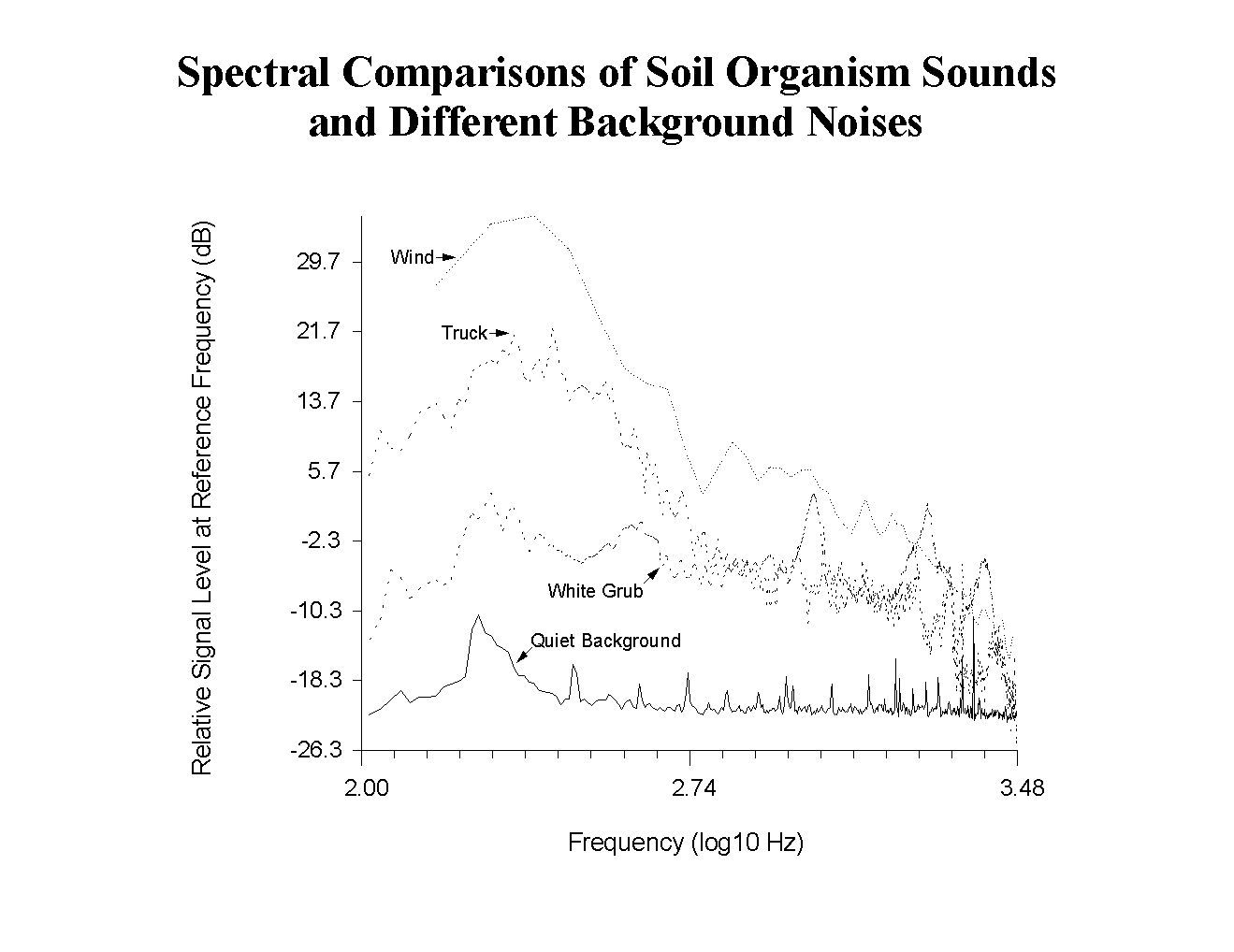
Figure 2. Spectral comparisons of sounds recorded by soil microphone from white grubs, and reference spectra of wind, vehicle noise, and the quiet noise background.
Louder background noises caused by wind or motor vehicles were more difficult to distinguish because they also had significant signal components above 400 Hz. In general, the ratio of the power below 400 Hz to the power above 400 Hz was much higher for the background than for the Phyllophaga signals, and the signal processing program could still distinguish these sounds automatically. Sounds made by other soil organisms could be distinguished from background for the same reason. Spectral templates for sounds made by millipedes, earthworms, mole crickets, and ants are shown in Fig. 3 for comparison with
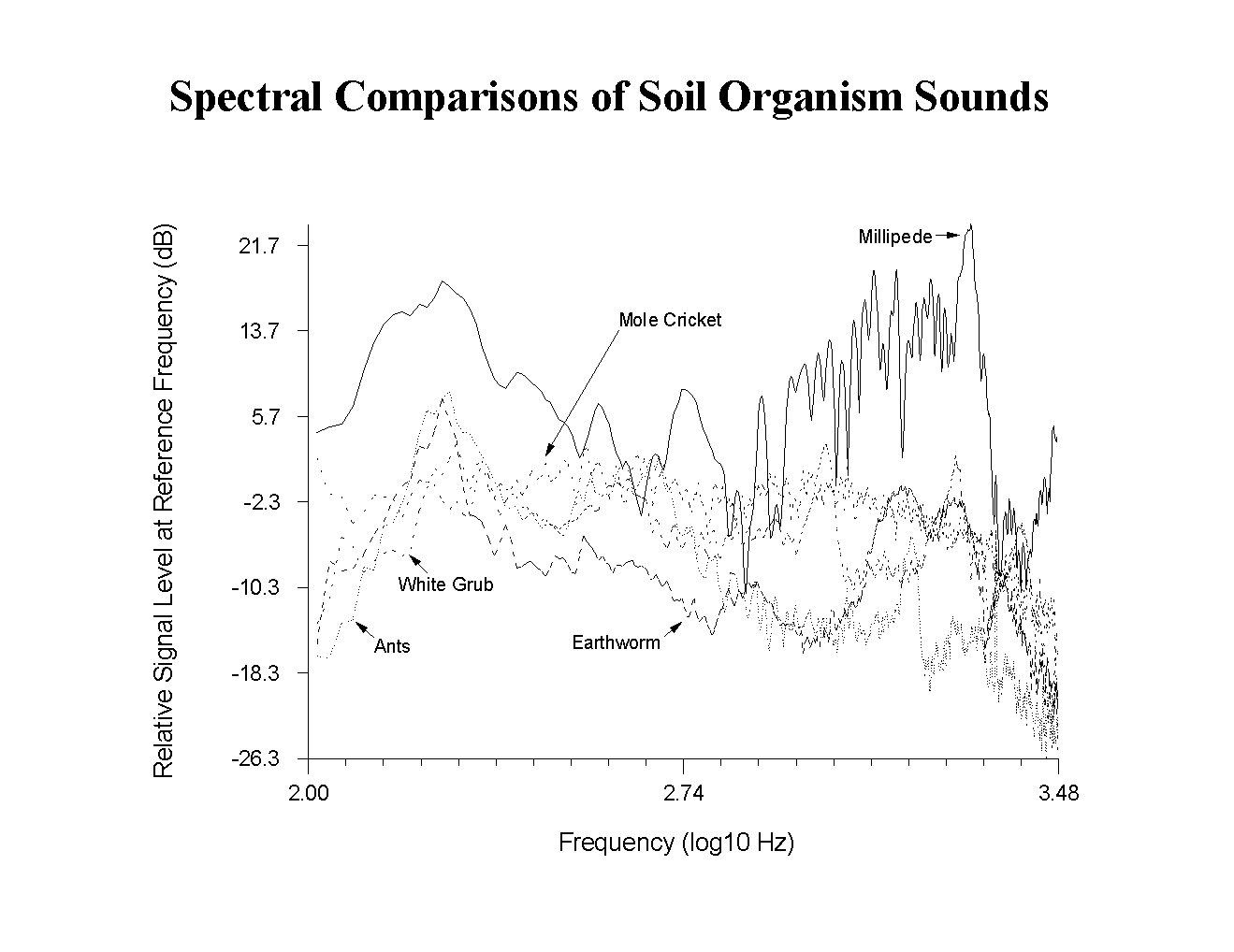
Figure 3. Spectral comparisons of sounds recorded by soil microphone from insects and other soil organisms commonly found in turf.
background noises and the Phyllophaga signals. The signals from these animals are so broadband that they cannot be distinguished from each other reliably. There is possibly a trend that the larger insects produce sounds with greater relative contributions above 1 kHz, but it is more difficult to distinguish these insects from each other than from background solely by using spectral templates. The lack of distinguishability is not surprising because all the sounds were made by an appendage digging in soil or scraping a root, and the animal was not using the sound for communication.
Anticipated Refinements and Improvements. A major need of turf, farm, and grove managers that would use an acoustic detection device is to distinguish between pest and nonpest organisms and to estimate the pest density. A possible solution to the problem of distinguishing different insect pests from nonpest soil organisms is to develop a dual microphone or accelerometer system that enables the location of the signal sources to be identified more precisely, and then to examine the temporal pattern of the signal at each location. Ants, in particular, produce a distinctive "patter" that may be distinguishable through temporal analysis. Earthworms often have a distinctive "slithering" pattern that can be distinguished by ear, and it may be possible to identify signal features that can distinguish these welcome organisms from pest insects.
A second benefit of a dual microphone/accelerometer system is that it allows more precision in the estimation of the number of insects within range of the sensor. Systems have already been successfully developed that count the number of hidden insects present in a sample of grain (7, 8), and it may be possible to adapt them for counting the number of insects present in a volume of soil. Experiments have already begun to estimate the total range over which acoustic sensors can detect insects of various sizes in soil. Preliminary experiments suggest that the range for a 100300 mg D. abbreviatus larva is 2050 cm.
We plan to continue testing with both systems because no clear favorite has yet emerged. The two systems have a similar range of detection. The accelerometer has a constant sensitivity between 03 kHz, while the soil microphone has low sensitivity below 0.5 kHz and peak sensitivity at > 0.9 kHz. The accelerometer system is more delicate than the soil microphone, but both systems are portable.
Longterm, we anticipate that an inexpensive, portable accelerometer or soil microphone system can be developed that pest managers and scouts will use as survey tools, reducing the necessity for laborintensive, destructive digging of samples. Trained listeners will be able to survey quickly for acoustic evidence of insects, and will be able to identify some insects by distinctive spectral and temporal patterns. Even when they cannot be identified precisely, the location of the sounds can be pinpointed so that the listener can directly sample the location if necessary.
ACKNOWLEDGMENTS
Everett Foreman, Eric Kaufmann, and John Green (Center for Medical, Agricultural, and Veterinary Entomology) assisted in recording and analysis of signals. Ian Jackson and Jerry Fojtik (Citrus Research and Education Center) assisted in field preparation and insect collection at Lake Alfred. This material is based partly upon work supported by the Cooperative State Research, Education and Extension Service, U. S. Department of Agriculture, under Agreement No. 58-6615-8-013. Funds for this project were also made available from the Citrus Production Research Marketing Order by the Division of Marketing and Development, Florida Department of Agriculture and Consumer Services, Bob Crawford, Commissioner.
REFERENCES
1. Crocker, R. L., RodriguezdelBosque, L. A., W. Nailon, W. T., and Wei, X. Southwest. Entomol. 21, 317324 (1996).
5. Mankin, R. W., J. Am. Mosq. Control Assoc. 10, 302308 (1994).
6. Mankin, R. W., Shuman, D., and Coffelt, J. A., J. Econ. Entomol. 90, 10321038 (1997).
7. Shuman, D., Weaver, D. K., and Mankin, R.
W., Appl. Acoust. 50, 279296. 1997.